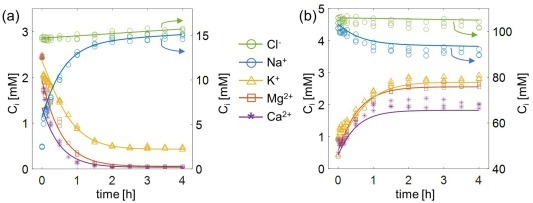
At the interface between an ion-exchange membrane and a multi-electrolyte solution, charged species redistribute themselves to minimize the free energy of the system. In this paper, we explore the Donnan equilibrium of membranes with quaternary electrolyte (Na+/Mg2+/K+/Ca2+/Cl−) solutions, experimentally. The data was used to calculate the ion activity coefficients for six commercial cation-exchange membranes (CEMs). After setting one of the activity coefficients to an arbitrary value, we used the remaining (N-1) activity coefficients as fitting parameters to describe the equilibrium concentrations of (N) ionic species with a mean relative error of 3 %. At increasing solution ionic strengths, the equivalent ion fractions of monovalent counter-ions inside the membrane increased at the expense of the multivalent ones in alignment with the Donnan equilibrium theory. The fitted activity coefficients were employed in a transport model that simulated a Donnan dialysis experiment involving all four cations simultaneously. The arbitrary value assigned to one activity coefficient affects the calculated Donnan potential at the membrane interface. Nevertheless, this arbitrary value does not affect the prediction of the ion concentrations inside the membrane and consequently does not affect the modeled ion fluxes. Read more here: https://www.sciencedirect.com/science/article/pii/S0021979724012645